Uterine Fibroids (UFs) (a.k.a. Uterine leiomyoma, myoma, fibromyoma, leiofibromyoma, fibroleiomyoma, and fibroma)
Overview
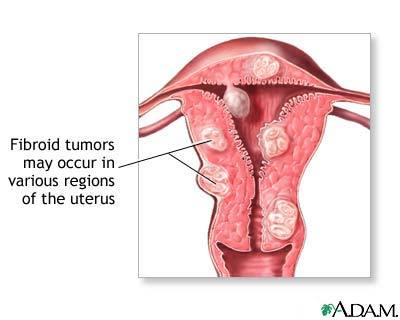
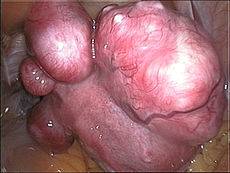
Uterine fibroids are BENIGN (i.e. non-cancerous), usually clusters of tumors in, on or within the uterine walls
Originate from and are composed of SMOOTH MUSCLE cells (myocytes) of the uterine wall’s muscle layer (called the myometrium, the middle layer of the uterine wall used for contracting the uterus) and its accompanying connective tissue;
- It is RARE for benign uterine leiomyomas to progress to cancerous leiomyosarcomas (1.7 women per 100,000 women are diagnosed annually with uterine sarcoma, which includes leiomyosarcoma) National Cancer Institute
Vary in size – from microscopic to very large (can weigh several pounds).
Symptoms of uterine fibroids
Not all UFs are symptomatic, but those that are can grow, causing:
- Heavy and painful menstruation; bleeding between periods; longer-lasing periods
- Painful sexual intercourse
- Urinary frequency and urgency
- Pelvic pain / pressure;
- Abdominal fullness, gas, constipation
- Pregnancy complications (rare) – increased blood flow and estrogen levels during pregnancy may cause UFs to grow, but return to normal size after delivery. Insufficient room in uterus may require early delivery; C-section may be needed if UFs block birth canal or cause wrong positioning of baby; may cause heavy bleeding immediately after giving birth.
Other complications of fibroids include:
- A pedunculated fibroid can become twisted and cause a kink in the blood vessels feeding the tumor – may need surgery;
- Anemia – may be severe with heavy bleeding
- Urinary tract infections – pressure from the fibroid can prevent bladder emptying fully;
- Malignant change (extremely rare) – called a leiomyosarcoma
- Infertility (rare)
Causes of uterine fibroids
Hormonal influences and growth factors are involved in UF growth and development
During a woman’s menstruating years, UFs typically continue to grow slowly
- Large fibroids may outgrow their blood supply and degenerate – described as hyaline, myxomatous, calcific, cystic, fatty, necrotic or red (usually only during pregnancy).
Fibroid growth seems to depend on both estrogen and progesterone hormones
It’s the “free” hormone levels that count. Some researchers maintain that serum estrogen and progesterone levels are unchanged by UFs – however, these serum levels are only meaningful if their free levels have been measured, which is not usually the case.
Overall effects of estrogen and progesterone:
- Mitogenic effect on leiomyoma cells. Encourages cell division / mitosis;
- Act by influencing (directly and indirectly) a large number of growth factors. Usually a protein or steroid hormone capable of stimulating cellular growth, proliferation and cellular differentiation (less specialized cell becomes a more specialized cell type).
Estrogen dominance. A dominance of estrogen over other hormones is a recognized problem of today, due to dietary and environmental changes.
Fibroids make a lot of estrogen, Fibroid cells can make their own estradiol and the conversion enzymes to make it are over-expressed in fibroids. Fibroids express higher levels of aromatase and can convert circulating androstenedione into estradiol via the enzymes aromatase and 17β-hydroxysteroid dehydrogenase Walker & Stewart, 2005; Shozu, 2004
- Aromatase over-expression in uterine leiomyoma tissue is particularly pronounced in African-American women (Ishikawa et al, 2009)
- Leptin (the “appetite suppressor” hormone) has also been shown to increase aromatase expression
Fibroids have a lot of Estrogen receptors
- Fibroid cells have more estrogen receptors (to respond to estrogen) than normal uterine muscle cells
- Having estrogen receptors, fibroids tend to enlarge during the reproductive years and shrink after menopause – In PREmenopausal fibroids the ER-β, ER-α (and progesterone) receptors are found over-expressed – compared to only ER-β in POSTmenopausal fibroids (which are rare) Strissel et al, 2007
A special ER-α genotype was found correlated with incidence and size of fibroids – Higher prevalence of this genotype in black women may also explain higher incidence of fibroids in Afro-American women. Most studies found that other different phenotypes in ER and PR gene encodings are not correlated with incidence of fibroids in Caucasian populations Alhendy, 2006
It is proposed that estrogen is growth-promoting by up-regulating:
- IGF-1, EGFR, TGF-β1 – Expression of transforming growth interacting factor (TGIF) is increased in leiomyoma compared with myometrium (In myometrial cells,TGIF is a potential repressor of anti-proliferative TGF-β pathways).
- Cytokines – signaling molecules secreted by nervous system glial cells and many immune system cells for intercellular communication.
- Apoptotic factors – TGF-β3 and PDGF, promotes aberrant survival of leiomyoma cells by down-regulating the tumor-suppressor protein p53.
Other hormones
Uterine fibroids have more progesterone receptors (to respond to progesterone) than normal uterine muscle cells.
- progesterone is thought to promote the growth of leiomyoma via up-regulation of EGF, TGF-β1 and TGF-β3
- progesterone is thought to promote the survival of leiomyoma via up-regulation of Bcl-2 expression and down-regulating TNF-α.
- progesterone is thought to counteract growth of leiomyoma by downregulating IGF-1.
A recent study emphasized the anomaly whereby >72% of women who were pregnant (or recently postpartum) have > 50% regression of pre-existing fibroids – One explanation points to the postpartum fall of progesterone.
Progesterone seems to have a dominant role by INCREASING mitotic rates in fibroids in the luteal phase of the menstrual cycyle (2nd half of cycle when corpus luteum secretes a lot of progesterone)– the drug mifepristone, a progesterone antagonist, INHIBITS fibroid growth lending support to progesterone’s dominant role. One theory is that progesterone upregulates EGF and TGF-β expression. However, progesterone also REDUCES the growth factor IGF-1 in vitro and INHIBITS MMPs, which activate growth factors and degrade extra cellular matrix (ECM), affecting ECM assembly and deposition, and so counters UF enlargement.
Other notes
- Actions of estrogen and progesterone are modulated by the “cross-talk” between themselves and prolactin – which controls the expression of their respective nuclear receptors.
- Rarely, leiomyomas progress to leiomyosarcomas and evolve to a hormone-non-responsive state – since many sarcomas have markedly reduced or no steroid hormone receptors. Alhendy A. & Salama S. (2006); Shozu M et al (2004); Strissel P et al (2007).
UF growth due to disorganized ECM with altered collagen fibers
- UFs have excessive production of a disorganized but stable extracellular matrix (ECM) and altered collagen fibrils in the ECM – fibroid collagen fibers (bunches of fibrils) are short, widely dispersed and lying non-parallel, compared to well-packed and lying parallel in the myometrium (smooth muscle tissue of the uterus).
- It is the abnormal and overproduced ECM that causes UF expansion , and not the slowly proliferating fibroid cells – UF tumors contain decreased/disrupted matrix metalloproteinases (MMPs) and more proteins in their ECM, such as collagen subtypes, proteoglycans, fibronectin, matrix glycoproteins and matricellular proteins (in particular thrombospondin-1 (TSP-1), which activates TGF- β and has a role in angiogenesis). The ECM binds cytokines and growth factors ready for action in the vicinity of the UF. Integrins are changed in UFs. The stability of this allbeit disorganized ECM requires therapeutic interventions that address ECM dissolution in addition to inhibiting cell proliferation
UFs involve growth factors:
Transforming Growth Factors-β1 and β3 (TGF- β1,TGF- β3) – have a central role in UF enlargement, in that they stimulate production/deposition of ECM and are acknowledged as important growth factors in fibrotic disease. E.g. Fibroids have more concentrated TGF-β receptors. Conversely, reduced TGF-β expression yields reduced ECM production and fibroid shrinkage
Other growth factors acting on myometrial (uterine smooth muscle) cells – epidermal growth factor (EGF), Insulin-Like Growth Factor (IGF), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF)
- Increased profibrotic cytokines (E.g. IL-1, IL-6, interferon, TNF- α) in UFs – involved with inflammatory response, cytokines are produced when growth factors act on target tissue.
UFs grow at different rates (even in the same woman) – and with different growth-rate patterns in white and African-American women
>50% of UFs are asymptomatic (i.e. have no symptoms) – ~70% of women by age 45 will be diagnosed with UFs, but only a fraction of those will cause problems or require treatment. Merck Manual
- Organochlorine pesticides stimulate leiomyomata cell proliferation in animals – organochlorines are xenoestrogens (i.e. mimic estrogen in the body). Hodges et al, 2000
Mainstream treatments for symptomatic uterine fibroids
General Treatments
- Oral contraceptives – to help control heavy periods
- Intrauterine devices(IUDs)that release the synthetic hormone progestin – to help reduce heavy bleeding and pain
- Iron supplements – to prevent or treat anemia due to heavy periods
- Nonsteroidal anti-inflammatory drugs (NSAIDs) – E.g.ibuprofen for cramps or pain
Surgical Treatments
- A hysterectomy is frequently advised in the U.S. – especially if a woman does not intend to have children. In fact, leiomyoma are the predominant reason for a hysterectomy in premenopausal women (MerckManual)
- Myomectomy – This surgery removes the fibroids. It is often the chosen treatment for women who want to have children, because it usually can preserve fertility. More fibroids can develop after a myomectomy.
- Magnetic Resonance-Guided Focused Ultrasound – Magnetic Resonance guided Focused Ultrasound (MRgFUS), is a non-invasive intervention (requiring no incision) that uses high intensity focused ultrasound (HIFU) waves to ablate (destroy) tissue in combination with Magnetic Resonance Imaging (MRI), which guides and monitors the treatment.
- Hysteroscopic resection of fibroids(as outpatient) – when UFs are growing inside the uterus. A small camera/instruments are inserted through the cervix into the uterus to remove the UFs.
- Uterine artery embolization – procedure cuts off blood supply to the UF, causing it to die and shrink.
Anti-fibrotic therapies inhibit and reverse the fibrotic process
Affect a change in abnormal ECM by leiomyoma cells
- Gonadotropin-releasing hormone analogues
- Small studies indicate progesterone receptor antagonists decrease uterine fibroid size
- Full progesterone receptor antagonist – Mifepristone (aka RU-486) was effective in a placebo-controlled pilot study. Malartic et al (2008)
- Selective progesterone receptor modulators – such as Progenta and Asoprisnil, have been under investigation; Wilkens et al, (2009)
Alternative treatments for uterine fibroids
Anti-estrogen therapies
High levels of estrogen are considered an element of uterine fibroids
Various tactics can be used to improve estrogen balance in the body:
How to reduce estrogen in the body
- Aromatase inhibitors used to reduce fibroids Malartic et al (2008) – The effect is believed to be partially due to (i) Lowering ovarian production and systemic estrogen levels and (ii) Inhibiting locally overexpressed aromatase in fibroids. Aromatase inhibitors have also been used experimentally in treatment of endometriosis, which indicated that aromatase inhibitors might be particularly useful in combination with a progestogenic ovulation inhibitor.
- Phytoestrogens – compete for receptors with endogenous estrogens; isoflavones daidzein and genistein are found in soy, but have been found to worsen fibroids when consumed in too high amounts. Lignans found in flaxseed.
- I3C in cruciferous vegetables – promotes formation of less potent estrogen metabolites Minich & Bland (2007)
- Reduce caffeine intake – to <500 mg / day (e.g. 2 cups coffee / day)
- Reduce alcohol consumption to 1 drink / day
- Increase fiber – helps remove excess estrogen from GI tract aiding excretion; reduces enterohepatic estrogen recirculation and/or shields estrogen absorption;
- Consume anti-inflammatory omega-3 fats and reduce inflammatory omega-6 fats to reduce estrogen production
- Iodine has a critical role in maintaining the body’s estrogen balance and can reduce uterine fibroids. Based on a controlled clinical trial with 1,365 women, 4mg daily of molecular iodine quickly resolves fibrocystic breast disease(FBD) – Iodine makes breast lumps and cysts disappear usually within only two months for most women. Iodine can similarly reduce uterine fibroids – one of the first conventional medical treatments for severe fibroids was to “paint”the uterus with iodine.
The primary aromatase promoter in leiomyomata tissues in non-Asian U.S. women is the inflammatory prostaglandin PGE2 Imir et al (2007)
Omega-3 fat reduces release of growth hormone – which promotes formation / growth of fibroids
Myoma is associated with beef and ham consumption, whereas high intake of green vegetables seems to have a protective effect Chiaffarino et al (1999)
Iodine -“The Universal Medicine”
Vitamin D3 decreases fibroid cell size and disrupts the formation of fibroid muscle cells
Vitamin D3is typically deficient in many populations today – E.g. Elderly, office workers, African Americans
- D3 treatment has been shown to inhibit leiomyocyte proliferation at physiological doses – Leiomyomas widely express the vitamin D receptor.
- Vitamin D decreases mitogenic activity of insulin and IGF-1
- Active metabolites of calcitriol (Active form of Vitamin D) down-regulate epidermal growth factor receptors (EGFRs) known to be active in mitogenic pathways in uterine leiomyomas. Down-regulation of these receptors shown to decrease growth / differentiation of tumor cells
- Risk of developing uterine fibroids in American black women REDUCED with just increased daily servings of “vitamin D-added” milk.
- Research shows that physiological doses of vitamin D have significant growth-inhibiting effect on leiomyomata cells Blauer et al (2009)
Vitamin D – “The Sunshine Vitamin”
Bioflavonoids reduce risk of malignant tumors
There is an inverse association between bioflavonoid intake and risk of malignant tumors – reported biological activities include:
- Induce apoptosis
- Cell cycle arrest
- Antiproliferative
- Anti-inflammatory
- Antioxidant protection against oxidative stress
- Anti-estrogenic
Asian women consume a lot of bioflavonoids and have lower incidence of hormonally dependent solid tumors – E.g. breast cancer in Asian women is 4-6 times lower than in American women, and several generations after migration to America they line up with the American statistics, suggesting an environmental rather than a genetic influence. Asian women consume a lot of soy-based foods, containing bioflavonoids that show up in blood and urine samples at significantly elevated levels.
“Bioflavonoids are . . . found in legumes, nuts, onions, apple, broccoli, red wine, grreen tea, cocoa powder, and dark chocolate. The best known anti-tumor flavonoids are epigallocatechin gallate (EGCG)from green tea, genistein (from soy and red clover), curcumin (from turmeric), silibin (from milk thistle), quercetin (from many yellow vegetables such as onions), and resveratrol (from grapes and red wine).”
– James H. Segars, author of the book Fibroids
Quercetin, EGCG, Curcumin, Silibrinin – (In berries, tea, grapes, olive oil, dark chocolate, walnuts, citrus):
- Inhibits IGF-1 signaling
- Anti-estrogenic – Estrogen receptor antagonist
- Alters cell cycle
Resveratrol (In red wine, grape, berries, dark chocolate, also peanuts (not recommended because of common fungal content); produced in plants in response to injury or fungal/ bacterial presence)
- Induces apoptosis
- Anti-estrogenic -Weak estrogen receptor agonist / antagonist
- Decreases collagen production
Resveratrol study-reported properties include:
- Anti-proliferative – mainly as a preventative; Decreases growth and increases death of fibroid cells in vitro;
- Cardio-protective
- Anti-diabetic
- Neuroprotective -possibly by chelating copper
- Reduces opioid tolerance -by increasing NMDA receptors
- Anti-inflammatory
- Antiviral activity
Curcumin (spice) – decreases growth and increases death of fibroid cells in vitro. Curcumin inhibited uterine leiomyoma cell proliferation by inducing apoptosis, and inhibited production of the ECM component fibronectin. (Malik et al, 2009; Kenji et al, 2011; )
Licorice (contains flavonoid isoliquiritigenin) – decreases growth and increases apoptosis of fibroid cells in vitro.
Green Tea (epigallocatechin gallate) – decreases growth of fibroid cells in vitro.
Retinoic acid
GI tract health strongly linked to uterine fibroid growth
Gastrointestinal problems (e.g. leaky gut syndrome, candida (yeast), intestinal bacterial overgrowth and gut inflammation) can indirectly lead to:
- Abnormal growth factor expression
- Excess estrogen
- Immune dysfunction.
- Toxic heavy metals can lead to abnormal bacterial growth in the gut and breakdown of the mucosal lining in the intestines. Hedburg, N (2011)
Female support herbs
There is supportive evidence thatvitex, yarrow and capsella buras-pastoris can reduce menstrual bleeding and PMS symptoms.
Vitex agnus-castus (Vitex, Chaste Tree, Chasteberry)
- Typical extract doses significantly inhibit prolactin secretion – (basal and TRH-stimulated) – presumed to be via dopaminergic effects. At low doses, such as might have been used in previous centuries for suppression of sexual desire, it inhibits activation of DOPAMINE 2 receptor by competitive binding, causing a slight increase ▲ in release of prolactin. In higher concentrations, as in modern extracts, the binding activity is sufficient to reduce ▼ the release of prolactin. A study found that treatment of 20 healthy men with higher doses of Vitex agnus-castus was associated with a slight reduction of prolactin levels, whereas lower doses caused a slight increase as compared to doses of placebo. (Merz et al, 1996)
A decrease of prolactin influences levels of follicle-stimulatin hormone (FSH) and estrogen in women, and testosterone in men.
- Chemical analysis of vitex agnus-castus has isolated the following compounds – flavonoids, alkaloids, diterpenoids, Vitexin, Casticin and steroidal hormone precursors, some of which are believed to affect the pituitary gland explaining its effects on hormone levels.
References
Alhendy, A.; Salama, S. (2006). “Ethnic distribution of ESTROGEN receptor-αpolymorphism is associated with a higher prevalence of uterine leiomyomas in black Americans”. Fertility and Sterility 86 (3): 686 PubMed
Baird DD, Dunson DB, Hill MC, et al. (2003) High cumulative incidence of uterine leiomyoma in balack and white women: ultrasound evidence. Am J Obstet Gynecol; 188: 100-107
Baird DD, et al. (2007) Association of physical activity with development of uterine leiomyoma. Am. J. Epidemiol. (2007) 165 (2): 157-163. Study
Blauer M, Rovio PH, Ylikomi T, Heinonen PK. (May 2009) Vitamin D inhibits mypmetrial and leimyoma cell proliferation in vitro. Fertility and Sterility. 91(5):1919-1925
Chiaffarino et al (Oct 1999) Diet and Uterine Myomas, Obstetrics and Gynecology 94(3):395-8 PubMed
Flake GP, Andersen J, Dixon D. (Jun. 2003) Review Etiology and pathogenesis of uterine leiomyomas: a review. Environ Health Perspect. Environ Health Perspect.111(8):1037-54. PubMed
Hedberg,Nikolas (2011) Renew Your Health Naturally Link
Hodges LC, Bergerson JS, Hunter DS, Walker CL. (2000 Apr) Estrogenic effects of organochlorine pesticides on uterine leiomyoma cells in vitro. Toxicol Sci. ;54(2):355-364. PubMed
Imir AG, Lin Z, Yin P, et al, (May 2007) Aromatase expression in uterine leiomyomata is regulated primarily by proximal promotors 1.3/II/. J. Clin Endocrinol Metab.; 92(5):1979-1982.
Ishikawa, H.; Reierstad, S.; Demura, M.; Rademaker, A. W.; Kasai, T.; Inoue, M.; Usui, H.; Shozu, M. et al. (2009). “High Aromatase Expression in Uterine Leiomyoma Tissues of African-American Women”. Journal of Clinical Endocrinology & Metabolism 94 (5): 1752. PubMed
Malartic, C.; Morel, O.; Akerman, G.; Tulpin, L.; Desfeux, P.; Barranger, E. (2008). “La mifépristone dans la prise en charge des fibromes utérins”. Gynécologie Obstétrique & Fertilité 36: 668. Link
Malik M , Mendoza M Payson M, Catherino W.H. (May 2009) Curcumin, a nutritional supplement with antineoplastic activity, enhances leiomyoma cell apoptosis and decreases fibronectin expression Fertility and Sterility. Volume 91, Issue 5, Supplement , Pages 2177-2184, Abstract
Merz, PG; Gorkow C, Schrödter A, Rietbrock S, Sieder C, Loew D, Dericks-Tan JS, Taubert HD (1996). “The effects of a special Agnus castus extract (BP1095E1) on prolactin secretion in healthy male subjects”. Exp Clin Endocrinol Diabetes 04 (6): 447-53. Link
Minich DM, Bland JS (June 2007) A Review of the clinical efficacy and safety of cruciferous vegeatable phytochemicals. Nutrition Revei ws.; 65(6):259-267.
Kenji Tsuiji et al (July 2011) Inhibitory effect of curcumin on uterine leiomyoma cell proliferation.Gyn. Endocrinolgy, Vol. 27, No. 7 , Pages 512-517 Abstract
Shozu, M.; Murakami, K.; Inoue, M. (2004). “Aromatase and Leiomyoma of the Uterus”. Seminars in Reproductive Medicine 22 (1): 51. PubMed
Strissel, P.; Swiatek, J.; Oppelt, P.; Renner, S.; Beckmann, M.; Strick, R. (2007). “Transcriptional analysis of steroid hormone receptors in smooth muscle uterine leiomyoma tumors of postmenopausal patients”. The Journal of Steroid Biochemistry and Molecular Biology 107(1-2): 42-47. . PubMed
Walker CL, Stewart EA (June 10, 2005) Uterine fibroids: the elephant in the room. Science. 308. PubMed pdf
Wilkens, J; Williams, AR; Chwalisz, K; Han, C; Cameron, IT; Critchley, HO (2009). “Effect of asoprisnil on uterine proliferation markers and endometrial expression of the tumour suppressor gene, PTEN”. Human reproduction (Oxford, England) 24(5): 1036-44 PubMed
Wise, L., Palmer, J., Bernard, H., Stewart, E., Rosenberg, L., (2005) Age-Specific Incidence rates for Self-Reported Uterine Leiomyomata in the Black Women’s Health Study Obstet Gynecol 105(3): 563-568